R&D report card: Zolgensma-maker Novartis' spending reaches $9.4B in 2019 | S&P Global Market Intelligence

Novartis' Zolgensma expansion hits FDA roadblock, giving Biogen and Roche a reprieve | Fierce Pharma

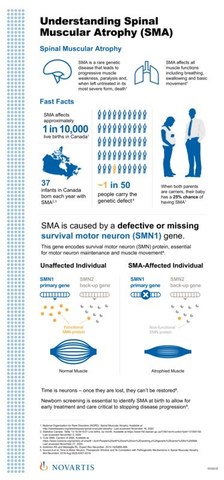
Health Canada approves Zolgensma®, the one-time gene therapy for pediatric patients with spinal muscular atrophy (SMA)

R&D report card: Zolgensma-maker Novartis' spending reaches $9.4B in 2019 | S&P Global Market Intelligence

/cloudfront-us-east-2.images.arcpublishing.com/reuters/T6XZTIM52JONFIX43C4EHTWNTQ.jpg)