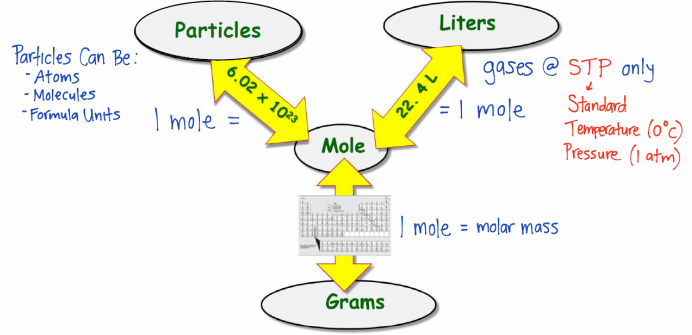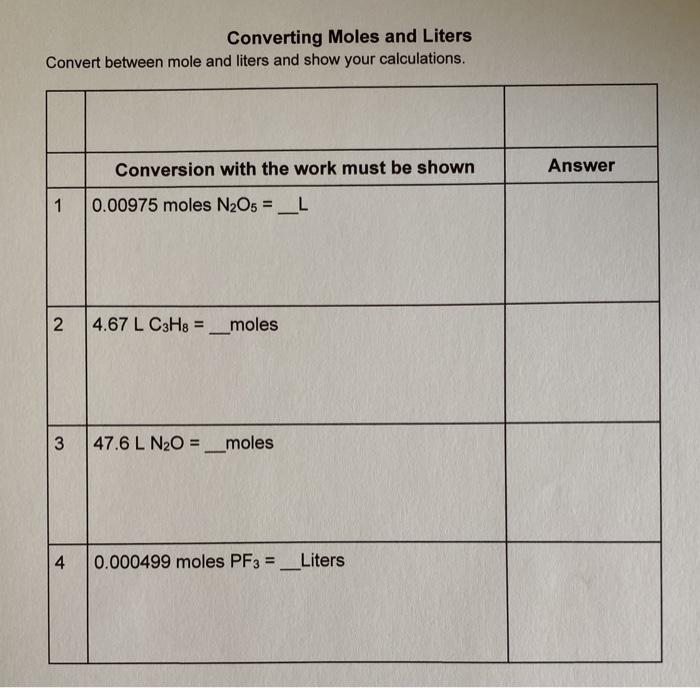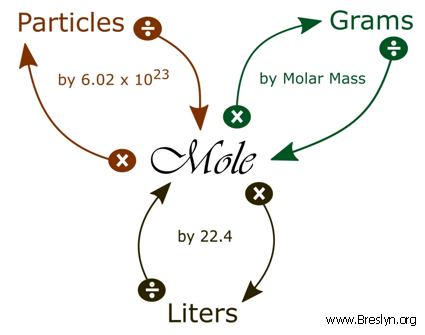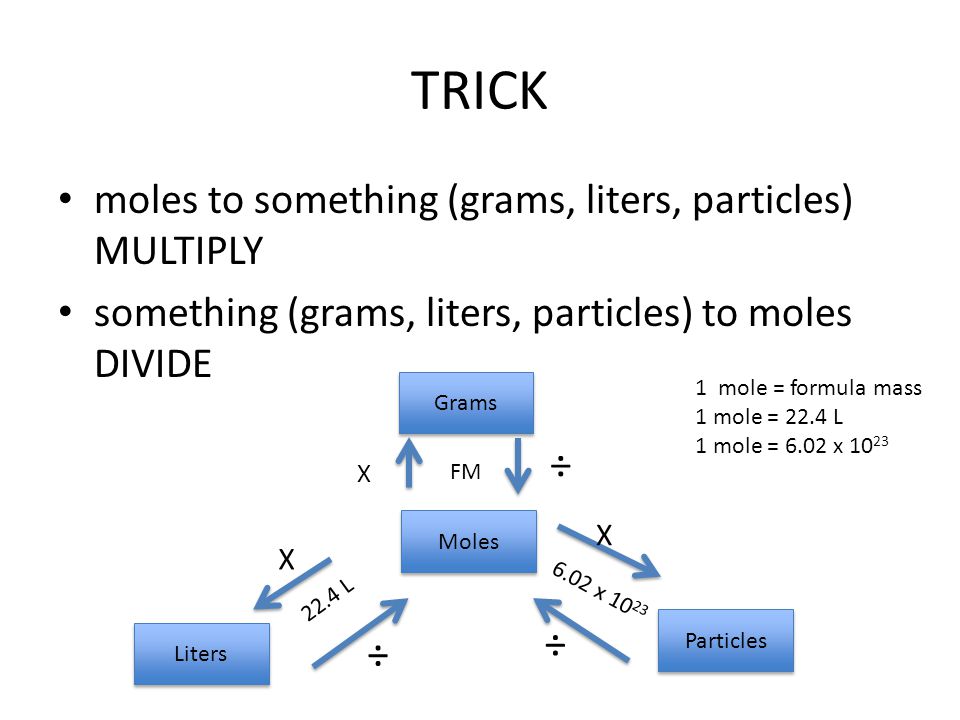
Converting Moles to Grams/Liters/Molecules/Atoms… Do Now: Convert the following What is the conversion factor for a)feet to inches: 1 foot = ___inches. - ppt download

Answer Key Moles & Liters volume .docx - Moles & Liters Volume We've established that it's not practical to count atoms so we often need to use | Course Hero

Liters To Moles Conversion: How to Convert Liters to Moles - by Wayne Breslyn | Mole conversion, Mole, Skin care solutions
In chemistry, why do we have to convert Liters, to moles, to grams, instead of just doing a one-step conversion (liters to grams)? - Quora
![Grade 10 honors chemistry] How come in moles to particles equations, the particle units are on the numerator where as in Particles to Moles (and other conversions) the particles are on the Grade 10 honors chemistry] How come in moles to particles equations, the particle units are on the numerator where as in Particles to Moles (and other conversions) the particles are on the](https://preview.redd.it/grade-10-honors-chemistry-how-come-in-moles-to-particles-v0-glehkjkeswg81.jpg?width=640&crop=smart&auto=webp&s=bcc0e9b6371de1b3a47732497ec6356d1b209d4e)
Grade 10 honors chemistry] How come in moles to particles equations, the particle units are on the numerator where as in Particles to Moles (and other conversions) the particles are on the





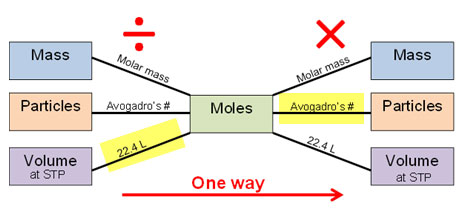




:max_bytes(150000):strip_icc()/606823-calculate-molarity-of-a-solution-FINAL-5b7d7e15c9e77c0050355d4e.png)