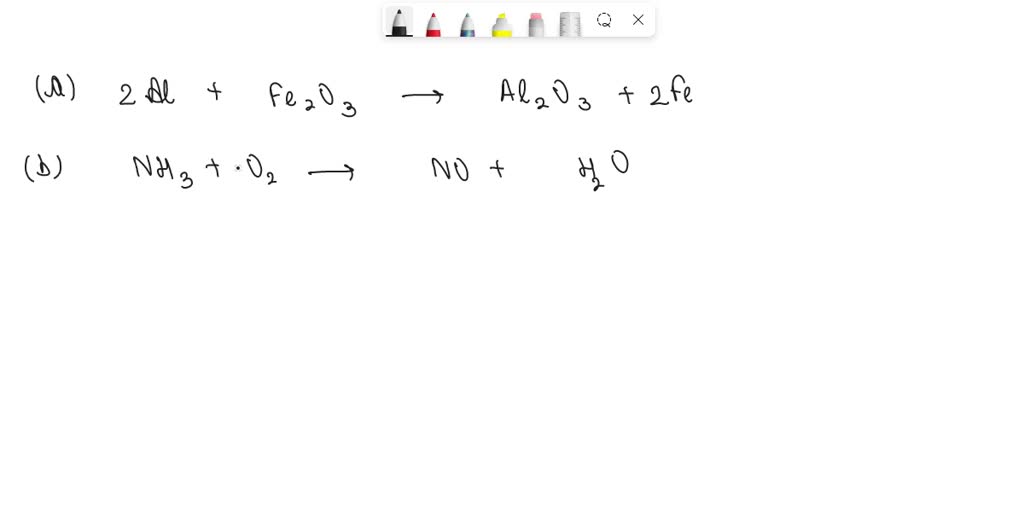
39. Fe2O3 + 2Al —> Al2O3 + 2Fe Which of the statement about the reaction below are incorrect? (|) Fe is getting reduced. (||)Al2O3 is getting oxidised (

SOLVED: Using the balanced equation 2 Al + Fe2O3 = Al2O3 + 2 Fe If you reacted 2.4 moles of aluminum with Fe2O3 and obtained 99.7 g of Al2O3, what would the percent yield of the reaction be?
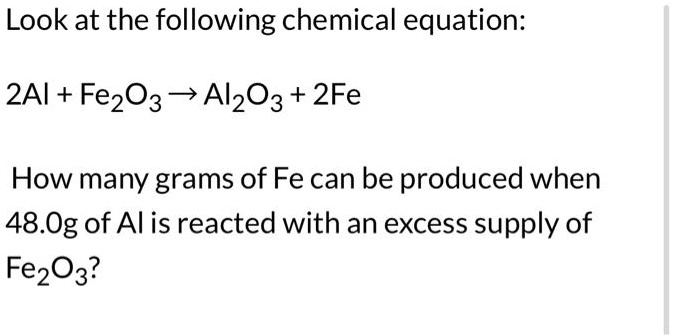
SOLVED: Look at the following chemical equation: 2Al + Fe2O3 -> Al2O3 + 2Fe How many grams of Fe can be produced when 48.0g of Al is reacted with an excess supply of Fe2O3?

Al + Fe2O3 → Al2O3 + Fe (Need to balance equation) How many grams of Fe can be produced when 10.0g of Al - brainly.com